DATING TECHNIQUES [0]
Rocks and other inanimate objects
Moving on from the basic principles underlying natural selection and its molecular counterpart, it is time now to appraise the techniques for dating rocks and other materials which have played a role in revealing the evidence for evolutionary process long after the events of which they speak have occurred.
Radiometric dating (also called radioactive dating) is a technique used to date materials such as rocks, usually based on a comparison between the observed abundance of a naturally occurring radioactive isotope and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth itself, and can be used to date a wide range of natural and man-made materials.
One such dating tool is the sensitive high resolution ion microprobe (SHRIMP), a large-diameter, double-focusing secondary ion mass spectrometer (SIMS) sector instrument housed in Canberra with about fourteen counterparts elsewhere in the world. A primary beam of ions[1] is focused on a sample, sputtering secondary ions which are then focused, filtered and measured according to their energy and mass. These secondary ions are measured with a mass spectrometer to determine the elemental, isotopic, or molecular composition of the surface.
The concept of the half-life
In brief, the atoms of a given element comprising the rocks are subject to radioactive decay and metamorphose into another element. The process starts when the rock, once molten lava, solidifies in the form of, let’s say, potassium-40 crystals which then decay into the radioactive isotope argon-40. When a crystal is newly formed, at the moment when molten rock solidifies, there is porassium-40 but no argon. The clock starts at this point, because there are no argon atoms in the crystal. In any sample of radioactive material, it always takes the same amount of time for half the sample to decay. This is referred to as the element's half life, and "this steady, reliable rate of decay (can) be used as a kind of clock". By calculating backwards from how much radiation a material has now and how swiftly it is decaying , you can work out its age.[2]
So when half the potassium atoms have decayed into argon-40 - a period of 1.26 billion years - this constitutes potassium's “half-life”. This half-life continues to decay at the same rate, so by measuring the ratio of potassium-40 to argon-40 in a piece of rock, geologists can get an idea of how long ago a rock first crystallised out of its molten state. By way of comparison, the half-life of rubidium-87 is 49 billion years and the half-life of fermium-244 is 3.3 milliseconds, which gives some idea of the range involved.
Here are some other examples[3]:
Unstable isotope Decays to Half-life (years)
Thorium-232 Lead-208 14,000,000,000
Uranium-238 Lead-206 4,500,000,000
Potassium-40 Argon-40 1,260,000,000
Uranium-235 Lead-207 704,000,000
Samarium-147[4] Neodymium-143 108,000,000
Iodine-129 Xenon-129 17,000,000
Aluminium-26 Magnesium-26 740,000
Carbon-14 Nitrogen-14 5,730
Isotopes are variants of a particular chemical element and represent the various configurations of its atoms. While all isotopes of a given element share the same number of protons, each isotope differs from the others in its number of neutrons. The number of protons within the atom's nucleus uniquely identifies an element, but a given element may in principle have any number of neutrons. The number of nucleons (protons and neutrons) in the nucleus, known as the mass number, is not the same for two isotopes of any element. Radioactive isotopes have unstable nuclei that decay, emitting alpha, beta, and sometimes gamma rays. Such isotopes eventually reach stability in the form of non-radioactive isotopes of other chemical elements, termed radiogenic daughters. In this context, unstable means that the atoms spontaneously decay into something else at a predictable rate, though not at predicable moments.
[0] See also http://elwynsbigbangpage.weebly.com/radioactivity-and-radioactive-decay.html
[1] An ion is a charged atom or molecule. It is charged because the number of electrons does not equal the number of protons in the atom or molecule. An atom can acquire a positive charge or a negative charge depending on whether the number of electrons in an atom is greater or less then the number of protons in the atom. When an atom is attracted to another atom because it has an unequal number of electrons and protons, the atom is called an ION. If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons, it is a positive ion.
Source: http://www.qrg.northwestern.edu/projects/vss/docs/propulsion/1-what-is-an-ion.html
[2] Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, 110.
[3] Source for Table and for the explanation in the preceding paragraph: Dawkins, Greatest Show 102.
[4] It has recently been suggested, based on “newer, more accurate technique” that this figure for Samarium may not be correct and should in fact be something more like 68 million years, meaning that every rock dated by Samarium-147 formed 20 to 80 million years earlier than thought – a lot for a system just 4.5 billion years old. Similarly with the pair of uranium isotopes which decay into lead. Sediments dated by these uranium isotopes are now thought to be younger by 700,000 years: “Ages of oldest rocks are off by millions of years”. New Scientist, 7 April 2012, p 9.
About carbon
Carbon has three isotopes: carbon-12, carbon-13 and carbon-14 with mass numbers (the combined numbers of protons and neutrons in the nucleus) 12, 13 and 14 respectively, making them three different varieties of the same chemical element, whose nuclei contain the same number of protons (six), but a different number of neutrons (six, seven and eight, respectively). (Electrons don’t figure because they have negligible mass). Almost 99% of the CO2 in the atmosphere contains the less heavy form of carbon: carbon 12, which is non-radioactive. A small part, 1.1% of CO2, is somewhat heavier, since it contains carbon 13 which is too short lived to worry about.
By comparison with carbon-12, carbon-14 represents a rather miniscule proportion of the carbon in the atmosphere, perhaps one part in a trillion, and is radioactive and unstable, since it decays into nitrogen 14 at a predictable rate. When an animal or plant dies and its body is severed from the food chain, it stops taking in new carbon. The ratio of carbon-12 to carbon-14 at the moment of death is the same as every other living thing, but from that moment on, the carbon-14 decays and is not replaced. The clock stops there, as Dawkins says.
The carbon-14 decays with its half-life of 5,730 years, meaning that the ratio of carbon-14 to carbon-12 in it decreases by half repetitively and regularly over successive periods of 5,730 years, while the amount of carbon-12 remains constant in the sample. Eventually, after say about 10 half-lives, it will all be carbon-12, or more strictly, the carbon-14 content will become too small to measure.
In the meantime, the ratio of carbon-12 to carbon-14 can be used to calculate the time that has elapsed since the death of the creature cut off from the food chain, and the age of other deceased organic substances such as wood, leather, cloth, and their interchange with the atmosphere. So by looking at the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.[1].
How carbon-14 is formed [2]
The significance for carbon dating
Because the half-life of carbon-14 is 5,730 years, it is only reliable for dating objects up to about 60,000 years old, but the same principles apply to other isotopes as well. For specimens older than about 50,000 to 60,000 years, carbon dating is unreliable, since the carbon-14 content will no longer be capable of measurement and we need to turn to a ‘slower’ clock: another radioactive isotope with a longer half-life.
Carbon dating techniques have been used to date human fossils found in the Lake Mungo World Heritage site in south western NSW at some 40,000 years. In conjunction with the technique known as mass spectrometry, it has also been used to date the shroud of Turin. It is accurate only to the nearest century or so, which is not too bad considered over a span of 50,000 years or thereabouts[4]. Carbon dating also has a number of other shortcomings which has caused scientists to turn to other techniques such as thermo-luminescence, which measure electrons trapped in clays, as an alternative.[5]
In 2014, the radioactive decay of uranium atoms contained in a substance called calcite or ‘cave popcorn’ covering cave paintings in a cave in the Indonesian island of Sulawesi has enabled their dating to a minimum age of between 39,900 to 17,400 years old, using a technique that measures the ratio of uranium and thorium. This has important implications for theories of human evolution, rock art being one of the first indicators of an abstract mind, since it places the paintings among the earliest known examples of cave art in the world. It was previously thought that Western Europe (esp Spain) was the centerpiece of early human artistic activity[6]. Such techniques were of course unknown in Darwin’s time. The fact that we can use them today only serves to make Darwin’s case on geological stratification and the timing of fossil finds so much the stronger.
[1] Source for explanation: Dawkins, Greatest Show, 103 ff; http://science.howstuffworks.com/environmental/earth/geology/carbon-142.htm
[2] Ibid.
[3] A term indicating its kinetic energy – the energy it possesses by being in motion - usually described in electron volts.
[4] My comment, not Dawkins’. In Chapter 4, Dawkins also considers and explains the wood dating technique known as dendrochronology, based on the tree ring formations within tree trunks as they age and how this can be extrapolated back by daisy-chaining techniques over some 11,500 years.
[5] In particular, the decay constant is understated by about 3%, and samples can be easily contaminated with carbon from other sources: Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, 153-154.
[6] “Sulawesi cave paintings some of earliest human art: study”, SMH, 9 October 2014.
Moving on from the basic principles underlying natural selection and its molecular counterpart, it is time now to appraise the techniques for dating rocks and other materials which have played a role in revealing the evidence for evolutionary process long after the events of which they speak have occurred.
Radiometric dating (also called radioactive dating) is a technique used to date materials such as rocks, usually based on a comparison between the observed abundance of a naturally occurring radioactive isotope and its decay products, using known decay rates. It is the principal source of information about the absolute age of rocks and other geological features, including the age of the Earth itself, and can be used to date a wide range of natural and man-made materials.
One such dating tool is the sensitive high resolution ion microprobe (SHRIMP), a large-diameter, double-focusing secondary ion mass spectrometer (SIMS) sector instrument housed in Canberra with about fourteen counterparts elsewhere in the world. A primary beam of ions[1] is focused on a sample, sputtering secondary ions which are then focused, filtered and measured according to their energy and mass. These secondary ions are measured with a mass spectrometer to determine the elemental, isotopic, or molecular composition of the surface.
The concept of the half-life
In brief, the atoms of a given element comprising the rocks are subject to radioactive decay and metamorphose into another element. The process starts when the rock, once molten lava, solidifies in the form of, let’s say, potassium-40 crystals which then decay into the radioactive isotope argon-40. When a crystal is newly formed, at the moment when molten rock solidifies, there is porassium-40 but no argon. The clock starts at this point, because there are no argon atoms in the crystal. In any sample of radioactive material, it always takes the same amount of time for half the sample to decay. This is referred to as the element's half life, and "this steady, reliable rate of decay (can) be used as a kind of clock". By calculating backwards from how much radiation a material has now and how swiftly it is decaying , you can work out its age.[2]
So when half the potassium atoms have decayed into argon-40 - a period of 1.26 billion years - this constitutes potassium's “half-life”. This half-life continues to decay at the same rate, so by measuring the ratio of potassium-40 to argon-40 in a piece of rock, geologists can get an idea of how long ago a rock first crystallised out of its molten state. By way of comparison, the half-life of rubidium-87 is 49 billion years and the half-life of fermium-244 is 3.3 milliseconds, which gives some idea of the range involved.
Here are some other examples[3]:
Unstable isotope Decays to Half-life (years)
Thorium-232 Lead-208 14,000,000,000
Uranium-238 Lead-206 4,500,000,000
Potassium-40 Argon-40 1,260,000,000
Uranium-235 Lead-207 704,000,000
Samarium-147[4] Neodymium-143 108,000,000
Iodine-129 Xenon-129 17,000,000
Aluminium-26 Magnesium-26 740,000
Carbon-14 Nitrogen-14 5,730
Isotopes are variants of a particular chemical element and represent the various configurations of its atoms. While all isotopes of a given element share the same number of protons, each isotope differs from the others in its number of neutrons. The number of protons within the atom's nucleus uniquely identifies an element, but a given element may in principle have any number of neutrons. The number of nucleons (protons and neutrons) in the nucleus, known as the mass number, is not the same for two isotopes of any element. Radioactive isotopes have unstable nuclei that decay, emitting alpha, beta, and sometimes gamma rays. Such isotopes eventually reach stability in the form of non-radioactive isotopes of other chemical elements, termed radiogenic daughters. In this context, unstable means that the atoms spontaneously decay into something else at a predictable rate, though not at predicable moments.
[0] See also http://elwynsbigbangpage.weebly.com/radioactivity-and-radioactive-decay.html
[1] An ion is a charged atom or molecule. It is charged because the number of electrons does not equal the number of protons in the atom or molecule. An atom can acquire a positive charge or a negative charge depending on whether the number of electrons in an atom is greater or less then the number of protons in the atom. When an atom is attracted to another atom because it has an unequal number of electrons and protons, the atom is called an ION. If the atom has more electrons than protons, it is a negative ion, or ANION. If it has more protons than electrons, it is a positive ion.
Source: http://www.qrg.northwestern.edu/projects/vss/docs/propulsion/1-what-is-an-ion.html
[2] Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, 110.
[3] Source for Table and for the explanation in the preceding paragraph: Dawkins, Greatest Show 102.
[4] It has recently been suggested, based on “newer, more accurate technique” that this figure for Samarium may not be correct and should in fact be something more like 68 million years, meaning that every rock dated by Samarium-147 formed 20 to 80 million years earlier than thought – a lot for a system just 4.5 billion years old. Similarly with the pair of uranium isotopes which decay into lead. Sediments dated by these uranium isotopes are now thought to be younger by 700,000 years: “Ages of oldest rocks are off by millions of years”. New Scientist, 7 April 2012, p 9.
About carbon
Carbon has three isotopes: carbon-12, carbon-13 and carbon-14 with mass numbers (the combined numbers of protons and neutrons in the nucleus) 12, 13 and 14 respectively, making them three different varieties of the same chemical element, whose nuclei contain the same number of protons (six), but a different number of neutrons (six, seven and eight, respectively). (Electrons don’t figure because they have negligible mass). Almost 99% of the CO2 in the atmosphere contains the less heavy form of carbon: carbon 12, which is non-radioactive. A small part, 1.1% of CO2, is somewhat heavier, since it contains carbon 13 which is too short lived to worry about.
By comparison with carbon-12, carbon-14 represents a rather miniscule proportion of the carbon in the atmosphere, perhaps one part in a trillion, and is radioactive and unstable, since it decays into nitrogen 14 at a predictable rate. When an animal or plant dies and its body is severed from the food chain, it stops taking in new carbon. The ratio of carbon-12 to carbon-14 at the moment of death is the same as every other living thing, but from that moment on, the carbon-14 decays and is not replaced. The clock stops there, as Dawkins says.
The carbon-14 decays with its half-life of 5,730 years, meaning that the ratio of carbon-14 to carbon-12 in it decreases by half repetitively and regularly over successive periods of 5,730 years, while the amount of carbon-12 remains constant in the sample. Eventually, after say about 10 half-lives, it will all be carbon-12, or more strictly, the carbon-14 content will become too small to measure.
In the meantime, the ratio of carbon-12 to carbon-14 can be used to calculate the time that has elapsed since the death of the creature cut off from the food chain, and the age of other deceased organic substances such as wood, leather, cloth, and their interchange with the atmosphere. So by looking at the ratio of carbon-12 to carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.[1].
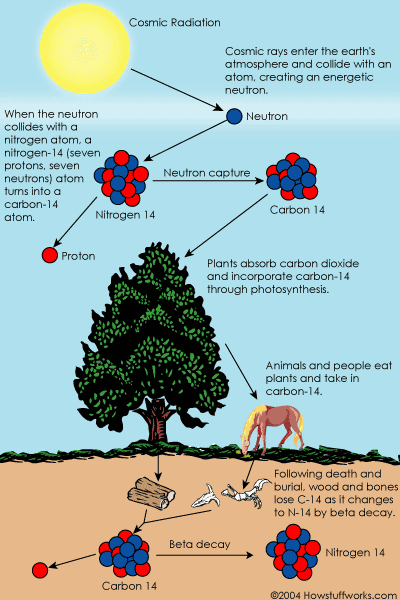
How carbon-14 is formed [2]
- Cosmic rays enter the earth's atmosphere. When one collides with an atom, an energetic neutron[3] is created. When one of these collides with a nitrogen atom, a nitrogen-14 atom with seven protons and seven neutrons turns into a carbon-14 atom with six protons and eight neutrons and a hydrogen atom having one proton only and no neutrons.
- The carbon 14 combines with oxygen to form carbon dioxide (CO2), which plants absorb naturally and incorporate into plant fibres via photosynthesis, the process whereby green plants take in carbon dioxide molecules from the atmosphere, using energy from sunlight to combine the carbon atoms with water to make sugars. All the carbon in ourselves and in all other living creatures comes ultimately, via plants, from carbon dioxide in the atmosphere, and it is continually being recycled back to the atmosphere: when we breathe out, when we excrete and when we die.
- When, animals and people eat plants, they naturally take in carbon-14. The ratio of normal carbon (carbon-12) to carbon-14 in the air and in all living things at any given time is nearly constant. Carbon-14 atoms are always decaying, but are being replaced by new carbon-14 atoms at a constant rate.
The significance for carbon dating
Because the half-life of carbon-14 is 5,730 years, it is only reliable for dating objects up to about 60,000 years old, but the same principles apply to other isotopes as well. For specimens older than about 50,000 to 60,000 years, carbon dating is unreliable, since the carbon-14 content will no longer be capable of measurement and we need to turn to a ‘slower’ clock: another radioactive isotope with a longer half-life.
Carbon dating techniques have been used to date human fossils found in the Lake Mungo World Heritage site in south western NSW at some 40,000 years. In conjunction with the technique known as mass spectrometry, it has also been used to date the shroud of Turin. It is accurate only to the nearest century or so, which is not too bad considered over a span of 50,000 years or thereabouts[4]. Carbon dating also has a number of other shortcomings which has caused scientists to turn to other techniques such as thermo-luminescence, which measure electrons trapped in clays, as an alternative.[5]
In 2014, the radioactive decay of uranium atoms contained in a substance called calcite or ‘cave popcorn’ covering cave paintings in a cave in the Indonesian island of Sulawesi has enabled their dating to a minimum age of between 39,900 to 17,400 years old, using a technique that measures the ratio of uranium and thorium. This has important implications for theories of human evolution, rock art being one of the first indicators of an abstract mind, since it places the paintings among the earliest known examples of cave art in the world. It was previously thought that Western Europe (esp Spain) was the centerpiece of early human artistic activity[6]. Such techniques were of course unknown in Darwin’s time. The fact that we can use them today only serves to make Darwin’s case on geological stratification and the timing of fossil finds so much the stronger.
[1] Source for explanation: Dawkins, Greatest Show, 103 ff; http://science.howstuffworks.com/environmental/earth/geology/carbon-142.htm
[2] Ibid.
[3] A term indicating its kinetic energy – the energy it possesses by being in motion - usually described in electron volts.
[4] My comment, not Dawkins’. In Chapter 4, Dawkins also considers and explains the wood dating technique known as dendrochronology, based on the tree ring formations within tree trunks as they age and how this can be extrapolated back by daisy-chaining techniques over some 11,500 years.
[5] In particular, the decay constant is understated by about 3%, and samples can be easily contaminated with carbon from other sources: Bill Bryson, A Short History of Nearly Everything, Broadway Books, 2003, 153-154.
[6] “Sulawesi cave paintings some of earliest human art: study”, SMH, 9 October 2014.
Dating fossils
Igneous rocks (rocks solidified from molten rock, underground magma in the case of granite, lava from volcanoes in the case of basalt, as opposed to being laid down in sediments) that overlie or underlie sedimentary strata, or are embodied in them, can be used to date those named sedimentary strata and hence the fossils within or between them: “between”, because fossils are not generally found in solidified igneous rocks, but rather in the softer sedimentary layers like limestone and sandstone which lie between. The disadvantage of this method is that we can’t exactly tell when the clock became zeroed in the case of sedimentary layers, typically layers of mud or silt or sand gradually laid down on the floor of a sea or lake or estuary, which then becomes compacted over the ages and hardens as rock. Corpses trapped in the mud then have the opportunity to fossilise and sedimentary rocks are the only kinds of rock that contain fossils worth speaking of. But we can use igneous rocks that overlie or underlie sedimentary strata, or are embedded in them to date those sedimentary strata and the fossils within them, because igneous rocks solidified more or less instantaneously upon their emergence as lava or magma, and we can then say with some degree of confidence, plus or minus one per cent) when the clock became zeroed.

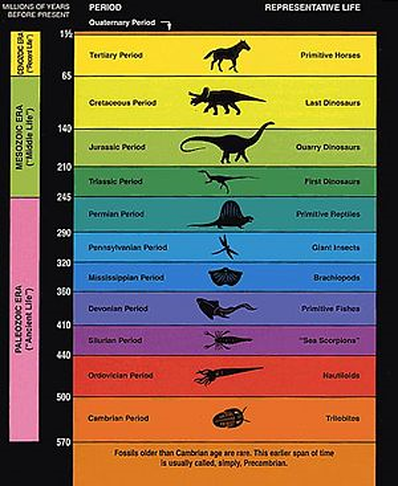
So, we can tell that our fossil is, say late Devonian, from its position in a Devonian stratum, and we know from the radioactive dating of igneous rocks found in association with the Devonian strata all around the world that the Devonian ended about 360 million years ago. (The principles of stratification and the naming of the various geological strata are considered later). What we can also say is that nothing one could remotely call a mammal has ever been found in Devonian rock or in any older stratum, Because of the age and slow rate of decay of these elements in igneous rock formations, they are referred to as “slow” radioactive clocks. Among the best-known techniques are radiocarbon dating, potassium-argon dating and uranium-lead dating. Geological timescales thus provide a significant source of information about the ages of fossils and the deduced rates of evolutionary change. They afford a reliable guide to the kind of creatures that inhabited the earth eons ago and when they did so. Dawkins concedes the possibility that at any moment, somebody might dig up a mammal in Cambrian rocks and the theory of evolution would be instantly blown apart. This is unlikely ever to happen, but the possibility that it could makes evolution a falsifiable, and therefore a scientific, theory [1]. The fossils which have been found in the different geological time zones are considered in the next section.
[1] Dawkins, Greatest Show, 100. As Dawkins and Wong also point out in The Ancestor's Tale - A Pilgrimage to the Dawn of Life, Weidenfeld and Nicolson, London, 2004, 2nd Edition, 2016, 88, the form of "sandwich dating" alluded to in the first paragraph is open to ther slight risk that a corpse has been carried by floodwater, or by hyenas or their dinosaur equivalents, to an anachronistic site. "With luck this will usually be obvious; otherwise we have to fall back on consistency with a general statistical pattern".
[1] Dawkins, Greatest Show, 100. As Dawkins and Wong also point out in The Ancestor's Tale - A Pilgrimage to the Dawn of Life, Weidenfeld and Nicolson, London, 2004, 2nd Edition, 2016, 88, the form of "sandwich dating" alluded to in the first paragraph is open to ther slight risk that a corpse has been carried by floodwater, or by hyenas or their dinosaur equivalents, to an anachronistic site. "With luck this will usually be obvious; otherwise we have to fall back on consistency with a general statistical pattern".
